Our Annual Stem Cell Research in Orthopedics Update—2016 Edition
Every year for the last few, I’ve created an infographic summarizing stem cell research in orthopedics. Every year, the list gets bigger and the number of patient results represented grows. This year is no different!
(Click here for a larger view)
How Is This Created?
I searched the US National Library of Medicine under several search terms around bone marrow concentrate and/or mesenchymal stem cell treatment under various orthopedic body areas looking for human studies. The only studies I excluded were case reports that were published later, after larger case series had already been reported. No attempt was made to try to figure out if any of the patients being reported were repetitive (some obviously were, and in some it was impossible to determine if the newer publication represented a new follow-up on a prior published set of patients or a new set).
How Does It Work?
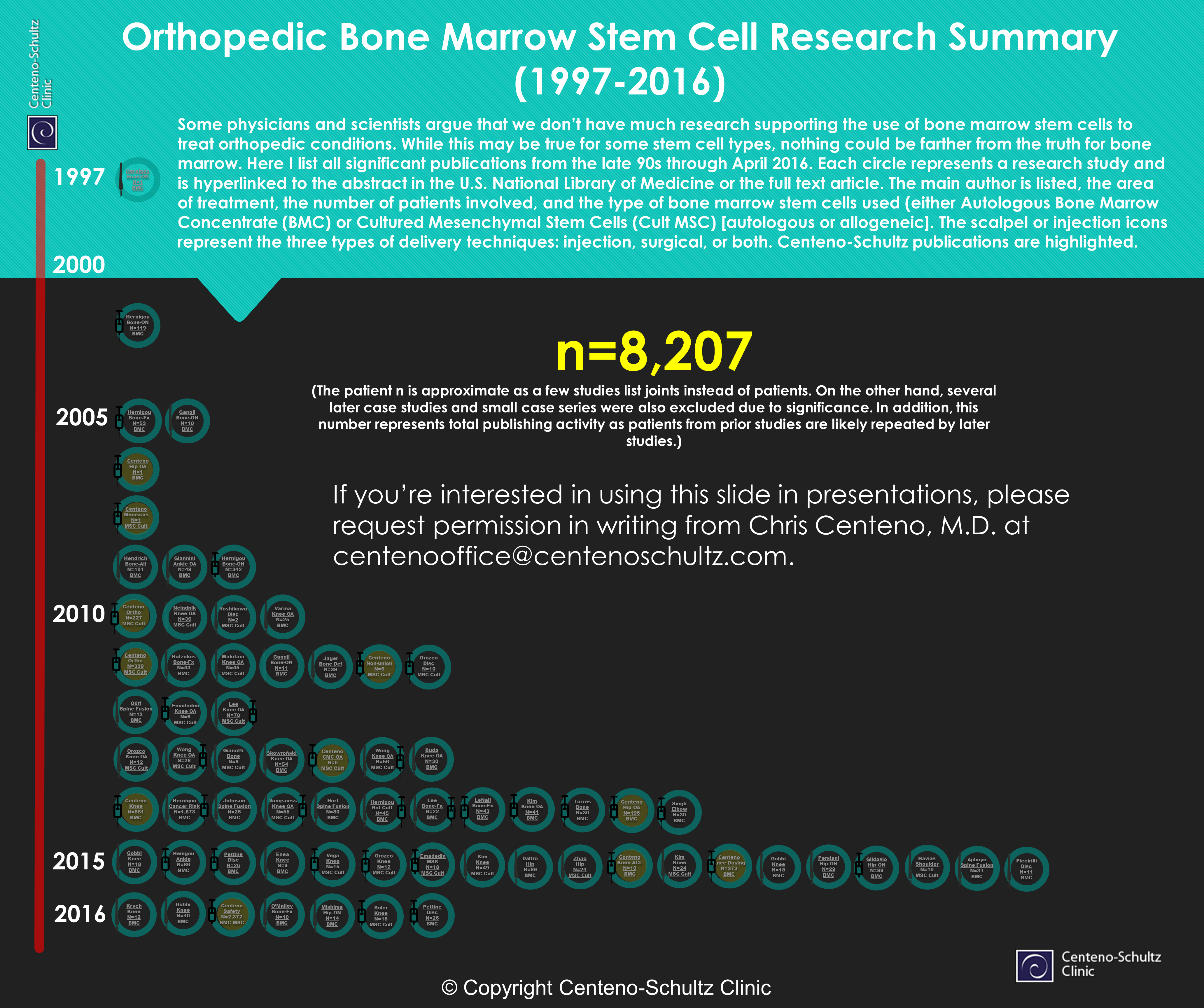
The infographic above is one massive PDF devoted to a summary document of stem cell research in orthopedics. Click on the picture above to be taken to that PDF. Each circle represents a research study and is hyperlinked to the abstract or the full text article in the US National Library of Medicine or elsewhere. The main author is listed as well as the area of treatment, the number of patients involved, and the type of bone marrow stem cells used (either autologous bone marrow concentrate [BMC] or cultured mesenchymal [cult MSC] [autologous or allogenic]). The scalpel or injection icons represent the three types of delivery techniques: injection, surgical, or both. Centeno-Schultz publications are highlighted.
What Does It Show?
The stem cell research in orthopedics is rapidly expanding. The total number of patients represented in this infographic increased from 5,513 last year to 8,207 this year. We now have two very large safety papers: Herigou’s n=1,873 paper, showing no safety issues around increased cancer rates, and our n=2,273 safety paper, reporting all adverse events and also showing an excellent safety profile. Reports of clinical outcome are mostly still small to large retrospective or prospective case series, but a few smaller randomized controlled trials are shown. Finally, almost every body part is represented: knee, hip, shoulder, ankle, lumbar spine, and hand.
What Are We Proud Of?
Regenexx patients now represent 51% of the total patient n reported on this document. That’s a testament to the fact that we are a research-based therapy and that we were the first in the U.S. to use this technology. In addition, we have always spend heavily tracking our patient outcomes. It’s also due to our great research team:
- The staff at more than 30 Regenexx sites across the U.S. that start the patient-registry process
- The Interventional Orthopedics Foundation staff that tracks patients
- The Regenexx Corporate biostats and clinical-research team
- The research writing team that we have assembled, including Dr. Freeman at OHSU, our annual fellows at the Centeno-Schultz clinic (hat tip to Dr. Pitts, Dr. Bashir, and Dr. Markle), and others.
Taking a rough idea for a registry-based study and refining it over months to something that can be submitted to a medical journal is an arduous and time intensive process. Hence a big thank you to all above!
Let’s Keep the Stem Cell Research in Orthopedics Tradition Going
This annual infographic is a labor of love, and as it gets bigger, it gets a little tougher to manage with each passing year. Last year a few kind souls let me know of a few broken or misdirected links, which were fixed. This year I’ve tried to check every hyperlink from scratch. However, if you find something that needs an edit or a study that should be on this one, don’t hesitate to reach out to me through my personal office e-mail at [email protected]. I have no issues sharing this with others who want to use it in medical-conference presentations—just drop me a line asking permission and I’ll be happy to allow it to be used (with an appropriate source credit).
The upshot? The stem cell research in orthopedics continues to look promising. This field is really the best of physician-based innovation—doctors seeing a problem on the ground and offering new solutions. In this type of innovation, physicians build the research base from scratch, which differs from the FDA/pharma route, where promising animal experiments get turned into clinical trials with high failure rates. While there are pros and cons to each type of innovation, I’m so thankful to be able to be a part of innovating in this field of orthopedic stem cells.

If you have questions or comments about this blog post, please email us at [email protected]
NOTE: This blog post provides general information to help the reader better understand regenerative medicine, musculoskeletal health, and related subjects. All content provided in this blog, website, or any linked materials, including text, graphics, images, patient profiles, outcomes, and information, are not intended and should not be considered or used as a substitute for medical advice, diagnosis, or treatment. Please always consult with a professional and certified healthcare provider to discuss if a treatment is right for you.