Stem Cell Viability 101 for Physicians

One of the most confusing areas for doctors and patients alike is the idea of what data indicates viable and functional stem cells in frozen umbilical cord and amniotic products. Most have no idea what to make of numbers thrown around by vendors, and many are easily confused. So if you’re a physician practicing in this space and don’t understand this topic, you are an easy mark for the scammers. Let me explain.
Understanding Cryopreservation
One of the problems with commercially available birth tissue products is that the initial viability of the cells is often in question. Meaning, there is a big difference between a birth product that is harvested from the OB ward and then brought to the lab to be immediately processed versus tissues that are saved in the hospital and then eventually shipped, processed, and frozen. As a rule, the longer the tissues are outside the body, the more cell death occurs.
In addition, when you freeze cells and then thaw them, this is a tremendous shock to the cells. Hence, properly frozen cells use certain strategies, like DMSO and controlled-rate freezing, to reduce ice crystal formation that can destroy cells. When these cells are thawed, they are often placed into an ideal cell-culture environment for days to make sure they recover. However, in the commercially available umbilical cord or amniotic products, the cells are shock-thawed in the hand and then injected.
These less-than-ideal conditions for sourcing and recovering cells from cryopreservation can result in lower initial viability numbers. For example, properly sourced, cryopreserved, and thawed cells have viability numbers that exceed 90%. However, when these numbers drop into the 80s, this generally indicates problems with cell health. When the numbers drop far lower, into what we often see with birth tissue products, such as viability numbers in the 40s to 70s, there are serious issues with cell health.
Levels of Viability Tests
Viability in the context of cells simply means what percentage of the cells are alive. There are four levels of viability tests that go from the least meaningful to the most important. Regrettably, the companies claiming to sell vials of live and functional stem cells often quote only the least meaningful.
These levels are:
- Simple live/dead dye exclusion stains
- Metabolic activity stains
- Apoptosis stains
- Cell culture assays
These tests can give wildly different results on the same sample. As an example, the first level of dye exclusion testing produces results that often don’t match up with the most meaningful test, a cell culture assay, giving a false positive viability test. To explain why, let’s review each test.
Simple Live/Dead Dye Exclusion Stains
The simplest test that can be performed to determine a snapshot of viability is a dye exclusion test. Here are the pros and cons:
Pros
- Simple to perform—results can be viewed under a light microscope and counted with an inexpensive hemocytometer
Cons
- Cells can be in the process of dying and still show positive on this test, so high false-positive rate
- Can produce more false positives based on dead tissue fragments excluding the dye
- Only a snapshot test—meaning, you can only look at whether cells are viable right now, which tells you little about how they will fare over time

The test is based on the principle that living cells have intact cell membranes that exclude certain dyes (such as trypan blue, eosin, or propidium), whereas dead cells tend to have broken membranes that will allow the dye into the cell. So dead cells stain and live cells do not.
The biggest problem that we’ve seen with dye exclusion stains is that it’s possible for dead tissue fragments to exclude the dye because they aren’t permeable to the dye. We’ve seen this with testing amniotic products that have fragments of the extracellar matrix from macerated and finely chopped membranes.
Metabolic Activity Stains
The next test up from a simple dye exclusion stain is one that relies on the metabolic activity of the cell. Here are the pros and cons:
Pros
- Two color stains—these stains often come in kits where, for example, live cells will turn one color and dead cells another contrasting color
Cons
- The more sophisticated metabolic activity stains involve either a flow cytometer or fluorescent microscope where the live cells fluoresce at a specific wavelength of light and the dead at another
- Cells can be in the process of dying but still show as alive because they have metabolic activity
- Only a snapshot test—meaning, you can only look at whether cells are viable right now, which tells you little about how they will fare over time
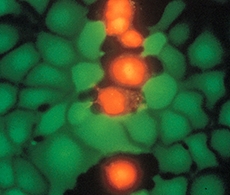
These tests usually work by taking advantage of natural cell enzymes to metabolize a chemical from colorless to colorful or fluorescent. Hence, unlike the dye exclusion tests, since they rely on metabolic activity to be positive, they are less likely to produce an immediate false positive. A commonly used test here is Sytox Red/Green.
Apoptosis Stains
The next step up is an apoptosis stain. Apoptosis is the process by which cells die. Here are the pros and cons:
Pros
- Can determine if a cell is alive but dying
Cons
- Only a snapshot test—meaning, you can only look at whether cells are viable right now, which tells you little about how they will fare over longer time periods. However, since you know the fraction of cells that are dying, this test reduces the false positive rate of a simple live/dead stain.
- Involves the use of either a flow cytometer or fluorescent microscope
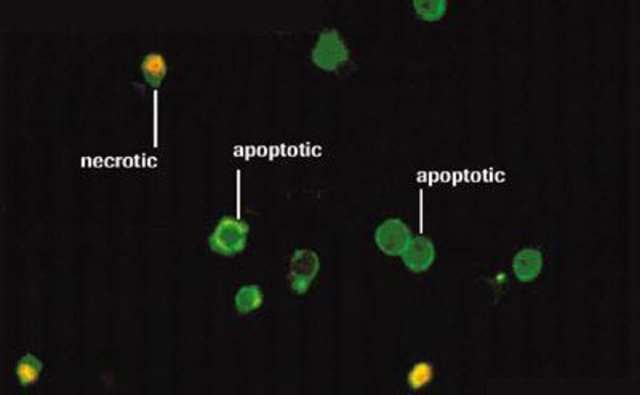
When cells go into preprogrammed cell death (apoptosis), they put up a marker on their surface (phosphatidylserine-PS) that identifies them to the clean-up machinery of the body. This apoptosis marker can be bound by a fluorescent antibody (usually annexin). Hence, this test can tell if the cell is alive but dying.
Cell Culture Assays
Finally, the most reliable way to test if you have viable and functional cells is a cell culture assay. These are the pros and cons:
Pros
- Can determine the number of cells that will survive under ideal conditions that mimic the body
Cons
- Cells then need to be identified via flow cytometry or through some other method
Here cells are placed in culture to determine how many will ultimately survive. Hence, this is the ultimate test of cell viability. However, the cells still need to be identified. For example, if the manufacturer claims mesenchymal stem cells, since they are adherent, one can simply rely on finding cells that attach to plastic. See my video below to see what this assay (also known as a CFU-f test) looks like. A second step would be identifying these cells via flow cytometry or differentiation assays.
The upshot? As a physician using these products and claiming that you’re injecting live and functional stem cells, it’s irresponsible to rely only on simple live/dead test results. That’s why you get paid the big bucks, to know the finer points of the science behind the wild claims of vendors selling products. Hence, please educate yourself. Right now, unless you have apoptosis testing and culture assays, relying on simple viability tests is a recipe for serious problems!

If you have questions or comments about this blog post, please email us at [email protected]
NOTE: This blog post provides general information to help the reader better understand regenerative medicine, musculoskeletal health, and related subjects. All content provided in this blog, website, or any linked materials, including text, graphics, images, patient profiles, outcomes, and information, are not intended and should not be considered or used as a substitute for medical advice, diagnosis, or treatment. Please always consult with a professional and certified healthcare provider to discuss if a treatment is right for you.